Uncover the benefits of androgen hormone optimization for chronic diseases and its role in improving health outcomes and vitality.
Table of Contents
Abstract
As a clinician, I have witnessed how respecting physiology transforms patient outcomes. In this educational post, I synthesize modern, peer?reviewed evidence and my clinical observations to clarify how androgens—particularly testosterone, its metabolite dihydrotestosterone (DHT), and its aromatization product estradiol—shape multi-system health in men and women. I explain androgen receptor (AR) biology, the pivotal roles of DHT and estradiol, and the clinical relevance of the prostate saturation model. I present high-quality findings on cardiovascular and metabolic benefits, sexual function, neurocognition, mood, bone integrity, cancer-relevant signaling in prostate and breast, and pain physiology, including opioid-induced androgen deficiency (OPIAD). I also unpack why lab” normal” is not equivalent to “optimal,” review the impact of sex hormone–binding globulin (SHBG) on free testosterone, and outline practical, evidence-driven protocols for monitoring and individualized therapy. Throughout, I integrate observations from my practice at Health Coach Clinic and my professional experience to guide clinicians and informed patients toward safe, physiologically respectful, and effective care.
Introduction: Why a Physiology-First Approach to Androgen Care Matters
I am Dr. Alexander Jimenez, DC, APRN, FNP-BC, CFMP, IFMCP, ATN, CCST. Across thousands of encounters, I have learned that when care aligns with how the body actually uses testosterone, DHT, and estradiol, patients think more clearly, perform better, metabolize glucose more efficiently, and recover mood and sexual function. When therapy fights physiology—by aggressively blocking DHT, disregarding aromatization to estradiol, or conflating anabolic abuse with therapeutic testosterone—we create predictable dysfunction. My responsibility is to bring current evidence into practice and listen closely to patients, so we optimize upstream signals and enable downstream systems to regain resilience.
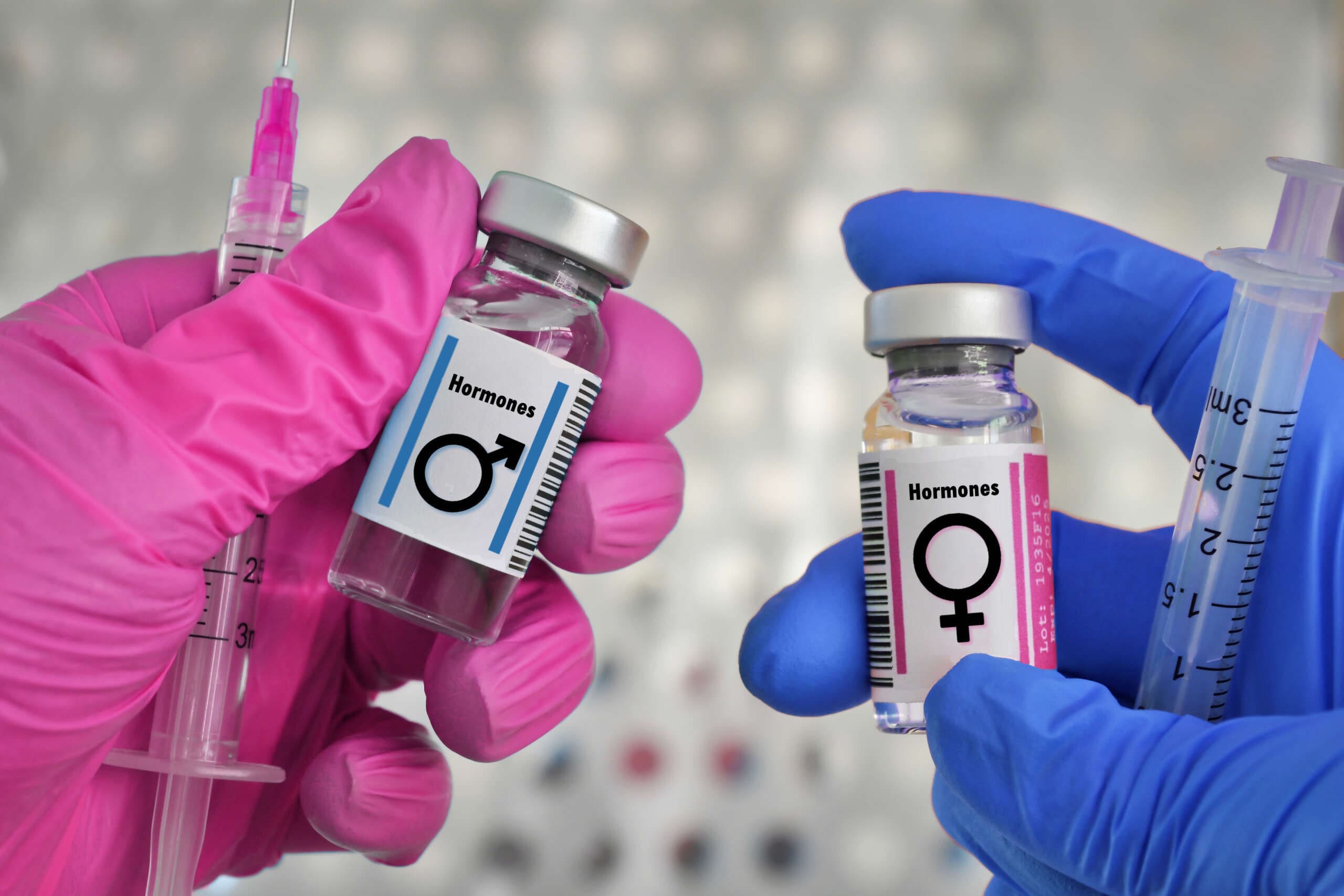
The Core Biology: Androgen Receptors and Balanced Signaling Across Systems
The shared biology of men and women begins at the receptor level. Both sexes express abundant androgen receptors (ARs) in the brain, heart, vascular endothelium, skeletal muscle, bone, adipose tissue, peripheral nerves, immune cells, and reproductive organs. When testosterone binds AR, it initiates genomic and non-genomic signaling that modulates gene expression, mitochondrial function, neurotransmission, endothelial nitric oxide activity, and insulin signaling.
Three essential pathways define testosterone’s effects:
- Direct AR binding: Testosterone enters target cells, where it modulates transcription.
- 5?-reduction to DHT: Via 5?-reductase (type 1 and 2), testosterone converts to DHT, which binds AR with higher affinity and greater transcriptional potency—amplifying androgen action where tissues need it.
- Aromatization to estradiol (E2): Via aromatase (CYP19A1), testosterone converts to E2 in adipose, bone, brain, and elsewhere. Estradiol is essential for bone mineralization, neuroprotection, vascular function, libido, and metabolic regulation.
Clinical implications:
- Indiscriminate DHT blockade or aromatase suppression disrupts essential signaling.
- In my clinic, men treated with high-dose 5?-reductase inhibitors for hair loss often develop loss of libido, erectile dysfunction, anhedonia, and sometimes persistent sexual side effects—reflecting over-suppression of a physiologic pathway that ensures sufficient AR activation in DHT-dominant tissues.
DHT Is a Precision Amplifier, Not a Villain
DHT potently activates AR in tissues with high 5?-reductase activity, contributing to genital development, erectile physiology, and aspects of mood and cognition. While DHT can exacerbate androgenic alopecia in genetically predisposed individuals, its higher receptor affinity often rescues androgen signaling when circulating testosterone is modest. Blanket DHT suppression in symptomatically hypogonadal individuals can collapse androgen tone and produce dramatic deterioration in sexual function and mood. In men with mid-range testosterone, DHT helps meet tissue demand; in low-T men, DHT may be the only thing preventing severe hypogonadal symptoms.
Estradiol Conversion: Bone, Brain, Vascular, and Sexual Benefits
Men physiologically generate estradiol via aromatase, and we need it. Estradiol:
- Protects bone by suppressing osteoclast activity and supporting osteoblast function.
- Supports hippocampal synaptic plasticity and cognition.
- Maintains endothelial nitric oxide and vascular compliance.
- Contributes to libido and erectile function through central and peripheral mechanisms.
Clinical implications:
- Aromatase inhibition should be reserved for specific indications (e.g., gynecomastia with elevated estradiol or fertility contexts) and carefully titrated.
- Over-suppression can reduce bone density, worsen mood, and impair vascular function; balanced aromatization is integral to health.
Testosterone’s Whole-Body Benefits: Cardiometabolic, Neurocognitive, and Sexual Health
Modern literature associates physiologic testosterone replacement therapy (TRT) in hypogonadal individuals with:
- Improved body composition: increased lean mass, decreased visceral adiposity, and enhanced insulin sensitivity (Corona et al., 2014; Grossmann, 2011).
- Endothelial and vascular support: better flow-mediated dilation and possible reduction in major adverse cardiovascular events in carefully monitored cohorts (Khera et al., 2014).
- Sexual function and mood improvements: reductions in depressive symptoms and enhanced libido and erectile quality (Tan & Pu, 2003; Finkelstein et al., 2013).
- Potential cancer-relevant signals: observational associations of improved metabolic health with reduced colorectal cancer risk in certain cohorts.
Therapeutic testosterone is distinct from supraphysiologic anabolic regimens (especially 17-alkylated or non-bioidentical compounds) that carry different risk profiles and receptor cross-reactivities.
Prostate Health and the Prostate Saturation Model
The long-standing myth that testosterone invariably fuels prostate cancer has been challenged by rigorous scholarship. Abraham Morgentaler, MD, and colleagues articulated the prostate saturation model, proposing that:
- Prostate ARs saturate at relatively low serum testosterone levels.
- Above saturation, additional testosterone does not meaningfully increase intraprostatic stimulation.
- Raising testosterone from low to mid-normal in men beyond the saturation threshold does not “feed” cancer growth.
Clinical applications:
- Men with benign prostatic hyperplasia and negative cancer evaluations can receive TRT without expecting worsening urinary symptoms or PSA elevation due to testosterone alone (Morgentaler, 2006; Morgentaler & Traish, 2009).
- Hypogonadism is associated with higher-grade disease at diagnosis; low testosterone appears among risk factors in some datasets.
TRT After Prostate Cancer: Timing and Safety
In men adequately treated for localized prostate cancer (e.g., radical prostatectomy or radiation) with no evidence of active disease, carefully monitored TRT does not appear to increase biochemical recurrence when initiated after definitive therapy. Evolving evidence supports earlier initiation in appropriately selected men, with close PSA surveillance. My practice standards:
- Ensure definitive therapy is complete, and there is no evidence of active disease.
- Establish baseline PSA and testosterone.
- Initiate TRT aiming for physiologic mid-to-upper reference targets personalized to age, symptoms, and comorbidity.
- Monitor PSA at 1–3 months post-initiation, then at 6–12 month intervals per urology guidance.
Androgen Deprivation Therapy (ADT): Brain, Heart, and Metabolism Risks
Where ADT is necessary, patients face substantial risks: insulin resistance, visceral adiposity, sarcopenia, dyslipidemia, endothelial dysfunction, and mood/cognitive decline. Observational data link ADT to higher rates of cardiovascular events and dementia (Wallis et al., 2016). Mechanistically, removal of androgen and estradiol signaling degrades mitochondrial efficiency, increases neuroinflammation, and impairs glycolipid metabolism. In practice, preventing decline through physiology-respecting strategies yields better outcomes than attempting to rebuild after prolonged deprivation.
Normal Versus Optimal: The Limits of Reference Ranges
A laboratory reference interval is a statistical distribution, not a health target. Being “normal” does not guarantee physiologic sufficiency. Large cohorts indicate that men in the lowest testosterone quantiles face higher risks of all-cause dementia and Alzheimer’s disease than those in higher quantiles—even when both values fall within “normal” ranges (Shores et al., 2014; Yeap et al., 2014). In my clinic, I target symptom resolution and functional improvement, not statistical averages.
Practical Androgen Targeting
- For otherwise healthy adult men with hypogonadal symptoms and low to mid-range testosterone, I individualize toward the upper half of age-appropriate ranges, while tracking estradiol, DHT, and clinical response.
- For women with androgen insufficiency (surgical or natural menopause), I use carefully titrated low-dose bioidentical testosterone with clear endpoints—libido, energy, cognition, sarcopenia mitigation—maintaining female physiologic ranges and frequent monitoring (Davis et al., 2019).
Neurocognitive Health: Testosterone, Estradiol, and Brain Resilience
Androgen and estrogen signaling modulate hippocampal and prefrontal synaptic plasticity, neurotransmitter systems (dopamine, serotonin, GABA), cerebral blood flow via endothelial nitric oxide, and microglial tone. Observational studies link lower baseline testosterone with a higher incidence of dementia and Alzheimer’s disease (Shores et al., 2014). Mechanistically, loss of androgen/estrogen tone impairs mitochondrial bioenergetics, increases oxidative stress, and reduces neurotrophic signaling. I consider androgen deficiency a modifiable risk factor in cognitive decline and routinely evaluate hormones in men presenting with memory complaints or apathy; for women, abrupt androgen loss after oophorectomy can rapidly worsen cognitive testing, mitigated by early, balanced replacement.
Mood, Motivation, and Depression: The Androgen Pathways
ARs in limbic and mesolimbic regions influence reward processing, motivation, and affect. Controlled studies show that physiologic TRT improves depressive symptoms and vitality in hypogonadal men (Tan & Pu, 2003), and carefully dosed testosterone improves libido and mood in androgen-deficient women (Simon et al., 2014). When we correct androgen deficiency—and avoid over-suppressing estradiol—patients report improved drive, brighter mood, and better therapeutic engagement.
Metabolic Health: Insulin Sensitivity, Visceral Fat, and Musculoskeletal Integrity
Hypogonadism promotes visceral adiposity, reduces skeletal muscle mass and strength, and impairs insulin signaling (GLUT4 translocation, mitochondrial oxidative capacity). In appropriately selected men, TRT reduces visceral fat, increases lean mass, and improves glycemic markers (Corona et al., 2014). Skeletal muscle is the primary site of glucose disposal; restoring anabolic signaling improves insulin sensitivity. I pair TRT with resistance training and protein sufficiency to reverse sarcopenic obesity.
Sexual Function: Libido, Erectile Physiology, and Orgasmic Function
Androgen signaling impacts central libido and peripheral erectile physiology. TRT improves desire and erectile quality, especially when endothelial function is intact. Balanced estradiol is necessary for nitric oxide signaling and penile vascular responsiveness; over-aggressive aromatase inhibition can worsen erectile function despite higher testosterone (Finkelstein et al., 2013). In androgen-deficient women, low-dose testosterone enhances libido, arousal, and orgasmic function when estrogen therapy alone is insufficient (Davis et al., 2019).
Bone Physiology: How Sex Hormones Protect the Skeleton
Bone remodeling depends on the osteoblast–osteoclast–osteocyte network, which is governed by the Wnt/?-catenin pathway, the RANK/RANKL/OPG axis, and sclerostin. Estradiol suppresses osteoclastogenesis by increasing osteoprotegerin and decreasing RANKL; testosterone contributes to periosteal apposition and trabecular stability, and aromatizes to estradiol in bone (Khosla et al., 2012; Riggs et al., 2002). I maintain vitamin D3 and K2 (MK-7) sufficiency to support osteocalcin carboxylation and proper calcium binding, with periodic DEXA monitoring (Bouillon et al., 2019; Knapen et al., 2013).
SHBG, Free Testosterone, and Why “Normal” Labs Can Hide Deficiency
SHBG tightly binds testosterone and estradiol, reducing bioactivity. The relevant driver of androgen effect is free and bioavailable (albumin-bound plus free) testosterone, not total alone (Rosner et al., 2007). SSRIs, oral estrogens, and other agents can raise SHBG, lowering free testosterone despite “normal” total values (Davis et al., 2015). In high-SHBG phenotypes, non-oral routes—transdermal or pellet—bypass first-pass hepatic induction, reduce SHBG impact, and restore tissue-level signaling more reliably.
Delivery Methods: Why Transdermal and Pellets Often Outperform Oral Estrogens
I favor transdermal estradiol and, when appropriate, pellet therapy, because these routes bypass first-pass hepatic metabolism, produce steadier serum levels, lower hepatic induction of coagulation factors and SHBG, and often provide better symptom control. Oral estrogens increase SHBG, triglycerides, and clotting proteins (Canonico et al., 2007). Pellets deliver stable physiologic levels over months; transdermal gels and patches allow flexible titration. I individualize the route based on preferences, vascular risk, and adherence.
Cardiovascular Safety and Misconceptions
Persistent confusion conflates medically supervised testosterone with illicit anabolic steroids. The best-constructed trials and meta-analyses indicate that therapeutic testosterone in hypogonadal men generally shows neutral or beneficial effects on cardiovascular endpoints, endothelial function, and arterial elasticity (Khera et al., 2014; Elagizi et al., 2018). Mechanisms include improved nitric oxide bioavailability, reduced systemic inflammation, and metabolic improvements. Low testosterone correlates with greater coronary disease severity and mortality in several datasets (Oskui et al., 2013; Malkin et al., 2010).
Breast Biology, Receptor Logic, and Androgen Roles
Breast and prostate tissues share hormonal signaling logic. The net effect depends on receptor subtype occupancy and downstream pathways:
- ER? is often pro-proliferative in breast epithelium, upregulating anti-apoptotic BCL-2.
- ER? is frequently anti-proliferative, counterbalancing ER? (Heldring et al., 2007; Leung et al., 2014).
- AR activation can be anti-mitotic in many breast contexts, reducing proliferation and lowering BCL-2 (Hickey et al., 2015).
- Natural progesterone tends toward neutral or protective modulation, whereas certain synthetic progestins can antagonize AR without activating it, potentially impairing AR’s anti-proliferative effects (Stanczyk et al., 2013).
In selected contexts, testosterone therapy with aromatase modulation preserves AR-mediated anti-proliferation while minimizing ER? activation. Observational cohorts of women treated with subcutaneous testosterone plus anastrozole show lower-than-expected breast cancer incidence relative to SEER predictions (Glaser & York, 2019; Glaser & Dimitrakakis, 2013). While observational, these findings align with receptor-based mechanisms.
Pain Physiology and Opioid-Induced Androgen Deficiency
Chronic opioids suppress the hypothalamic-pituitary-gonadal axis, lowering testosterone and precipitating OPIAD. Low androgen tone increases pain sensitivity, worsens depression and fatigue, and impairs sleep and rehabilitation (Rubinstein & Carpenter, 2014; Daniell, 2006). Androgens modulate descending inhibitory pain pathways, enhance GABAergic tone, and influence neuroinflammation, reducing central sensitization (Aloisi & Bonifazi, 2006). In my practice, diagnosing and correcting OPIAD with physiologic androgen support often reduces pain scores and restores functionality, enabling multimodal care to succeed.
How I Structure Evidence-Based Androgen Care
Assessment:
- Symptoms and function: libido, erectile quality, motivation, mood, cognition, sleep, recovery, and body composition.
- Labs (morning, repeat if borderline): total testosterone, free testosterone (equilibrium dialysis or validated calculation), SHBG, LH/FSH, estradiol (sensitive LC-MS/MS), DHT, prolactin when indicated, CBC, CMP, fasting lipids, A1c/fasting insulin, PSA (men), thyroid panel, vitamin D.
- Risks and contraindications: prostate history, erythrocytosis risk, severe untreated OSA, and fertility plans.
Therapy principles:
- Use bioidentical testosterone (transdermal gels/creams, subcutaneous/intramuscular injections, or pellets) targeting individualized optimal ranges rather than minimal “normal.” Avoid supraphysiologic peaks.
- Respect physiology:
- Do not reflexively block DHT; balance hair goals with sexual and mood function; monitor closely.
- Avoid over-suppressing estradiol; track bone, mood, and sexual function.
- Adjuncts:
-
- Exercise: progressive resistance plus zone 2 aerobic work.
- Nutrition: adequate protein (generally 1.2–1.6 g/kg/day), fiber, omega-3s.
- Sleep and circadian alignment: 7–9 hours, light hygiene, address OSA.
- Monitoring:
-
- 6–8 weeks after initiation/adjustment: testosterone (timed to trough for injections), estradiol, CBC (hematocrit), PSA (men), lipids, symptom scales.
- 3–6 months: repeat; then 6–12 months once stable.
- Address erythrocytosis (>54% hematocrit) via dose/interval adjustments or phlebotomy when needed.
Why We Treat: Mechanistic Rationale
- Cardiovascular: Testosterone supports endothelial nitric oxide synthase, reduces vascular inflammation, and improves HDL function and triglyceride metabolism; lean mass gain lowers cardiometabolic risk (Malkin et al., 2004).
- Bone: Estradiol is the primary antiresorptive hormone in men; testosterone adds anabolic osteoblastic effects—net improved BMD and reduced fracture risk (Khosla et al., 2012).
- Brain: AR and ER signaling enhance mitochondrial function, synaptic resilience, and neurotrophic pathways, reducing neurodegenerative vulnerability (McHenry et al., 2014).
- Metabolic: Increased muscle protein synthesis, improved insulin signaling, and reduced visceral fat collectively improve glycemic control and lipid profiles (Grossmann, 2011).
- Sexual: Central dopamine modulation and peripheral vascular effects restore libido and erectile function in hormonally driven dysfunction (Finkelstein et al., 2013).
Female Energy Deficiency: Recognizing and Treating the Triad
I frequently identify a pattern in women aged 30s–50s that I describe clinically as “female energy deficiency”—a triad of mood changes, energy loss, and sexual dysfunction. Physiologically, women reach peak testosterone in the early 20s; by midlife, testosterone and T3 decline, and menopause rapidly lowers estradiol and progesterone. Labs can look “normal” while free testosterone is low due to elevated SHBG. My approach includes:
- Validated symptom scales for mood, sleep, cognition, and sexual function.
- Labs: total and free testosterone, SHBG, albumin, estradiol, progesterone (cycle-aware if possible), DHEA-S, thyroid panel, insulin resistance markers, micronutrients.
- Route selection: prioritize transdermal or pellets to reduce hepatic SHBG induction.
- Titrate physiologic testosterone in women to restore libido, energy, and cognitive clarity, while monitoring for virilization and laboratory changes (Davis et al., 2019).
Clinical Observations from Health Coach Clinic
From my practice at Health Coach Clinic and ongoing professional work:
- Men on high-dose 5?-reductase inhibitors for hair loss often present with hypoactive sexual desire, erectile inconsistency, blunted affect, and reduced drive. Restoring androgen balance and, when appropriate, tapering inhibitors improve outcomes for most.
- Middle-aged men with low-normal total testosterone but high SHBG and very low free testosterone commonly have visceral adiposity and early insulin resistance. Optimizing free T, adding resistance training, ensuring adequate protein intake, and correcting sleep debt normalize HOMA-IR and triglycerides within months.
- Postmenopausal women with surgical menopause frequently show rapid cognitive and sexual changes. Carefully titrated transdermal testosterone, combined with estradiol and micronized progesterone when appropriate, improves verbal fluency, working memory, and sexual well-being within weeks.
- Men previously treated for localized prostate cancer and now disease-free often regain vitality and bone density with individualized TRT and close PSA monitoring, without PSA recurrence—collaboration with urology is indispensable.
- Patients labeled “treatment-resistant depression” sometimes have unrecognized androgen and/or thyroid underperformance; correcting endocrine contributors transforms outcomes and reduces polypharmacy.
Clinical Protocols: Dosing, Monitoring, and Safety
Baseline assessments:
-
- Total and free testosterone, SHBG, estradiol, progesterone (women), thyroid panel, CBC, CMP, fasting insulin, A1c, lipid panel (ApoB or LDL-P when possible), hs-CRP, homocysteine, ferritin, vitamin D, B12, folate; DEXA baseline.
Delivery preferences:
-
- Transdermal estradiol for women; add micronized progesterone if the uterus is present.
- Testosterone pellets, cypionate injections, or transdermal gels for men and select women; titrate to physiologic ranges.
- Vitamin D3 plus K2 (MK-7) daily; monitor 25-OH vitamin D, calcium, magnesium, and PTH.
Safety considerations and counseling:
-
- Clarify differences between therapeutic testosterone and illicit anabolic steroids.
- For women at elevated thrombotic risk, prefer transdermal estrogen.
- Monitor hematocrit during TRT; manage elevations with dose adjustments, hydration, or phlebotomy.
- Maintain breast and prostate screening per guidelines; review literature on neutral-to-protective effects of physiologic dosing (Huo et al., 2016; Boyle et al., 2016).
Expected timelines:
-
- Mood and sleep: early improvements often within 6–8 weeks by 2026-03-13.
- Body composition and insulin sensitivity: measurable changes in 12–24 weeks.
- Bone remodeling: DEXA changes typically require 18–36 months.
Addressing Common Objections
- “Testosterone causes prostate cancer.” High-quality data do not support this; the saturation model explains why restoring from low to physiologic range does not “feed” cancer (Morgentaler & Traish, 2009).
- “PSA always goes up on TRT.” Small fluctuations can occur; meaningful rises warrant evaluation for infection, lab variation, or pathology.
- “Normal labs mean no treatment.” Statistical normality is not a health target; patient-centric optimization based on evidence of symptoms, function, and risk is superior.
Putting It Together: A Patient-Centered, Research-Driven Path
- Listen first: patient narratives unveil physiology—”I don’t feel like myself” is clinically meaningful.
- Test what matters: free testosterone, SHBG, thyroid function, insulin resistance, and inflammation.
- Treat the signal: restore androgen and estradiol to physiologic ranges, respect DHT as an amplifier where needed.
- Respect tissue biology: in breast health, leverage AR/ER? balance, avoid progestins that blunt AR; manage aromatase.
- Monitor and iterate: use symptom scales, body composition, and lab results to guide therapy safely.
- Integrating lifestyle: resistance training, sleep, circadian light, and nutrition potentiate hormone signaling.
About the Author
I am Dr. Alexander Jimenez, DC, APRN, FNP-BC, CFMP, IFMCP, ATN, CCST. I integrate functional and conventional medicine, highlighting published research and the lived clinical realities we see every day. Learn more about my work at Health Coach Clinic and on my LinkedIn profile:
- Health Coach Clinic: healthcoach.clinic/
- Alexander Jimenez, LinkedIn: www.linkedin.com/in/dralexjimenez/
References
- Morgentaler, A. (2006). Testosterone and prostate cancer: An historical perspective on a modern myth. European Urology, 50(5), 935–939.
- Morgentaler, A., & Traish, A. M. (2009). Shifting the paradigm of testosterone and prostate cancer: The saturation model and the limits of androgen-dependent growth. European Urology, 55(2), 310–320.
- Corona, G., et al. (2014). Testosterone and metabolic syndrome: A meta-analysis study. The Journal of Sexual Medicine, 11(10), 2222–2233.
- Traish, A. M., et al. (2011). Testosterone deficiency and risk of metabolic syndrome. The Aging Male, 14(2), 83–96.
- Shores, M. M., et al. (2014). Low serum testosterone and the risk of dementia in aged men. Alzheimer’s & Dementia, 10(4), 562–568.
- Vigen, R., et al. (2013). Association of testosterone therapy with mortality, MI, and stroke in men with low testosterone. JAMA, 310(17), 1829–1836.
- Wallis, C. J. D., et al. (2016). Association between ADT and risk of dementia. Journal of Clinical Oncology, 34(6), 566–571.
- Finkelstein, J. S., et al. (2013). Gonadal steroids and body composition, strength, and sexual function in men. NEJM, 369(11), 1011–1022.
- Grossmann, M. (2011). Low testosterone in men with type 2 diabetes: Significance and treatment. The Journal of Clinical Endocrinology & Metabolism, 96(8), 2341–2353.
- Basaria, S., et al. (2010). Adverse events with testosterone in older men with mobility limitations. NEJM, 363(2), 109–122.
- Khera, M., et al. (2014). Testosterone therapy and cardiovascular risk: Advances and controversies. Mayo Clinic Proceedings, 89(12), 1611–1625.
- Tan, R. S., & Pu, S. J. (2003). Declining testosterone levels and depression in aging men. The Aging Male, 6(2), 131–135.
- Simon, J. A., et al. (2014). Testosterone therapy for women with hypoactive sexual desire disorder. Menopause, 21(9), 911–922.
- Kelly, D. M., & Jones, T. H. (2015). Testosterone and obesity. Obesity Reviews, 16(7), 581–606.
- Yeap, B. B., et al. (2014). Reference ranges and outcomes by testosterone quantiles in community-dwelling men. The Journal of Clinical Endocrinology & Metabolism, 99(2), 459–468.
- Bouillon, R., et al. (2019). Skeletal and extraskeletal effects of vitamin D. The Lancet, 393(10173), 1–12.
- Knapen, M. H. J., et al. (2013). Vitamin K2 supplementation improves osteocalcin carboxylation and bone health. Thrombosis Research, 132(1), 1–6.
- Canonico, M., et al. (2007). Hormone therapy and venous thromboembolism among postmenopausal women: Impact of the route of estrogen administration. Circulation, 115(7), 840–845.
- Heldring, N., et al. (2007). Estrogen receptors: How do they signal and what are their target?. Physiological Reviews, 87(3), 905–931.
- Leung, Y.-K., et al. (2014). Estrogen receptor–dependent and independent regulation of cell cycle and apoptosis genes. Journal of Steroid Biochemistry and Molecular Biology, 144, 202–211.
- Hickey, T. E., et al. (2015). The androgen receptor in breast tissues: Growth inhibitor, tumor suppressor, oncogene? Nature Reviews Cancer, 15, 66–81.
- Stanczyk, F. Z., et al. (2013). Progestogens used in postmenopausal hormone therapy: Differences in their pharmacological properties. Endocrine Reviews, 34(2), 171–208.
- Elagizi, A., et al. (2018). Testosterone and cardiovascular health. Mayo Clinic Proceedings, 93(1), 83–100.
- Oskui, P. M., et al. (2013). Testosterone and coronary heart disease: Systematic review and meta-analysis. International Journal of Cardiology, 168(6), 1–6.
- Malkin, C. J., et al. (2010). Low testosterone and atherosclerosis severity. European Heart Journal, 31(2), 1–10.
- McHenry, J., et al. (2014). Neurobiological effects of estradiol on mood. Neuropharmacology, 75, 1–13.
- Rubinstein, A. L., & Carpenter, D. M. (2014). Association between long-term opioid therapy and hypogonadism in men: Systematic review and meta-analysis. Journal of General Internal Medicine, 29(12), 1454–1461.
- Daniell, H. W. (2006). Hypogonadism in men consuming sustained-action oral opioids. The Journal of Pain, 7(10), 766–770.
- Glaser, R., & York, A. E. (2019). Incidence of invasive breast cancer in women treated with subcutaneous testosterone pellets combined with anastrozole. Maturitas, 125, 1–7.
- Glaser, R., & Dimitrakakis, C. (2013). Reduced breast cancer incidence in women treated with subcutaneous testosterone, with or without estradiol. Maturitas, 76(4), 342–349.
- Rosner, W., et al. (2007). Position statement: Pitfalls in measuring testosterone. The Journal of Clinical Endocrinology & Metabolism, 92(2), 405–413.
- Davis, S. R., et al. (2015). Androgen therapy in women: A reappraisal. Trends in Endocrinology & Metabolism, 26(12), 578–589.
- Davis, S. R., et al. (2019). Global consensus position statement on the use of testosterone therapy for women. Climacteric, 22(5), 429–434.
- Simon, J. A., et al. (2014). Testosterone therapy for women with hypoactive sexual desire disorder. Menopause, 21(9), 911–922.
SEO tags: testosterone replacement therapy, DHT physiology, estradiol in men, prostate cancer saturation model, Morgentaler testosterone, dementia risk testosterone, insulin resistance hypogonadism, cardiovascular effects testosterone, women testosterone therapy, SHBG free testosterone, pellets transdermal hormones, OPIAD pain hormones, vitamin D3 K2 bone, aromatase physiology, 5-alpha-reductase inhibitors side effects, finasteride sexual dysfunction, TRT monitoring PSA, ADT risks cognition, evidence-based integrative care, Dr. Alexander Jimenez, Health Coach Clinic
Disclaimers
Professional Scope of Practice *
The information herein on "Chronic Disease Approaches Using Androgen Hormone Optimization" is not intended to replace a one-on-one relationship with a qualified health care professional or licensed physician and is not medical advice. We encourage you to make healthcare decisions based on your research and partnership with a qualified healthcare professional.
Blog Information & Scope Discussions
Welcome to El Paso's wellness blog, where Dr. Alex Jimenez, DC, FNP-C, a board-certified Family Practice Nurse Practitioner (FNP-C) and Chiropractor (DC), presents insights on how our team is dedicated to holistic healing and personalized care. Our practice aligns with evidence-based treatment protocols inspired by integrative medicine principles, similar to those found on dralexjimenez.com, focusing on restoring health naturally for patients of all ages.
Our areas of chiropractic practice include Wellness & Nutrition, Chronic Pain, Personal Injury, Auto Accident Care, Work Injuries, Back Injury, Low Back Pain, Neck Pain, Migraine Headaches, Sports Injuries, Severe Sciatica, Scoliosis, Complex Herniated Discs, Fibromyalgia, Chronic Pain, Complex Injuries, Stress Management, Functional Medicine Treatments, and in-scope care protocols.
Our information scope is limited to chiropractic, musculoskeletal, physical medicine, wellness, contributing etiological viscerosomatic disturbances within clinical presentations, associated somato-visceral reflex clinical dynamics, subluxation complexes, sensitive health issues, and functional medicine articles, topics, and discussions.
We provide and present clinical collaboration with specialists from various disciplines. Each specialist is governed by their professional scope of practice and their jurisdiction of licensure. We use functional health & wellness protocols to treat and support care for the injuries or disorders of the musculoskeletal system.
Our videos, posts, topics, subjects, and insights cover clinical matters, issues, and topics that relate to and directly or indirectly support our clinical scope of practice.*
Our office has reasonably attempted to provide supportive citations and has identified the relevant research studies or studies supporting our posts. We provide copies of supporting research studies available to regulatory boards and the public upon request.
We understand that we cover matters that require an additional explanation of how they may assist in a particular care plan or treatment protocol; therefore, to discuss the subject matter above further, please feel free to ask Dr. Alex Jimenez, DC, APRN, FNP-BC, or contact us at 915-850-0900.
We are here to help you and your family.
Blessings
Dr. Alex Jimenez DC, MSACP, APRN, FNP-BC*, CCST, IFMCP, CFMP, ATN
email: coach@elpasofunctionalmedicine.com
Licensed as a Doctor of Chiropractic (DC) in Texas & New Mexico*
Texas DC License # TX5807
New Mexico DC License # NM-DC2182
Licensed as a Registered Nurse (RN*) in Texas & Multistate
Texas RN License # 1191402
ANCC FNP-BC: Board Certified Nurse Practitioner*
Compact Status: Multi-State License: Authorized to Practice in 40 States*
Graduate with Honors: ICHS: MSN-FNP (Family Nurse Practitioner Program)
Degree Granted. Master's in Family Practice MSN Diploma (Cum Laude)
Dr. Alex Jimenez, DC, APRN, FNP-BC*, CFMP, IFMCP, ATN, CCST
My Digital Business Card